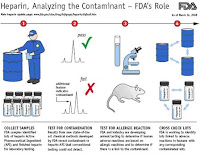
Yesterday on Capitol Hill, a house subcommittee heard testimony on the recent issue of contaminated Heparin. Robert Parkinson, CEO of Baxter testified that the company believes the heparin was deliberately tainted.
Yesterday on Capitol Hill, a house subcommittee heard testimony on the recent issue of contaminated Heparin. Robert Parkinson, CEO of Baxter testified that the company believes the heparin was deliberately tainted. Most heart wrenching, was when the family’s victims told their stories on how the tainted Heparin has destroyed their lives.
Leroy Hubely, was in tears as he explained how his wife Bonney died in December 2007 and then a month later his son, Randy died under the same circumstances. Only after the recall of Heparin, did he learn from his local dialysis center that tainted heparin, and not some cruel twist of fate, led to both of their deaths.
His daughter-in-law Collen Hubely, a dialysis nurse, testified: “As a nurse, I thought that I would be there to save my husband from any errors, but I guess I was naive. I never thought the life saving medication we were relying on might be contaminated. If citizens are truly going to ever to feel safe in this country going to the hospital, a doctor, taking your daily medication; we have a responsibility to be make sure that everything that is given is free from contamination.”
Families demanded more stringent policies and testing to protect consumers from dangerous materials hidden in our medications.